
Active sites are unique microenvironments. 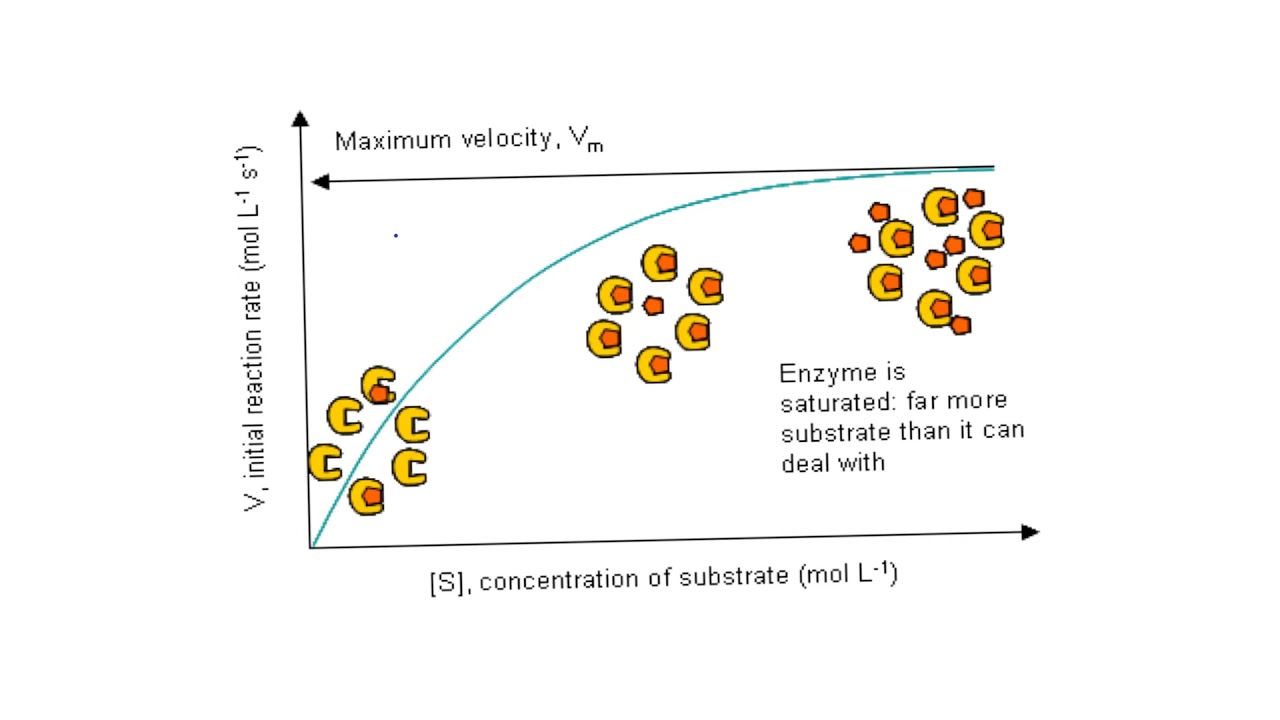
In many proteins the remaining amino acids also constitute regulatory sites, sites of interaction with other proteins, or channels to bring the substrate to the active sites.ģ. The "extra" amino acids serve as a scaffold to create the three dimensional active site from the amino acids that are far apart in the primary structure. Nearly all enzymes are made up of more than 100 amino acid residues. Most of the amino acid residues in an enzyme are not in contact with the substrate, which raises the question of why enzymes are so big. The active site takes up a relatively small part of the total volume of an enzyme. The active site is a three dimensional cleft or crevice formed by groups that come from different parts of the amino acid sequence - residues far apart in the amino acid sequence may interact more strongly than adjacent residues in the sequence.Ģ. 
Although enzymes differ widely in structure, specificity, and mode of catalysis a number of generalizations concerning their active sites can be made:ġ. These residues are called the catalytic groups. It contains the residues that directly participate in the making and breaking of bonds. Also, enzymes can be inhibited by specific molecules by called competitive, uncompetitive, and noncompetitive inhibitors.Ĭatalysis happens at the active site of the enzyme. Enzymes take advantage of the full range of intermolecular forces ( van der waals interactions, polar interactions, hydrophobic interactions and hydrogen bonding) to bring substrates together in most optimal orientation so that reaction will occur. The most remarkable characteristics of enzymes are their ability to accelerate chemical reactions and their specificity for a particular substrate. 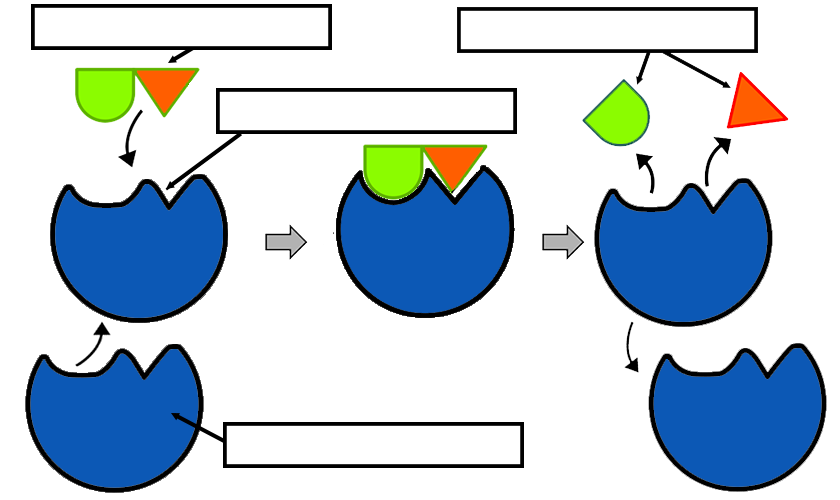
Virtually all enzymes are proteins, though the converse is not true and other molecules such as RNA can also catalyze reactions. Some biological reactions in the absence of enzymes may be as much as a million times slower. This is usually done by accelerating reactions by lowering the transition state or decreasing the activation energy. Enzymes are macromolecules that help accelerate (catalyze) chemical reactions in biological systems.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
